7 product results for:
"RFP Trap"
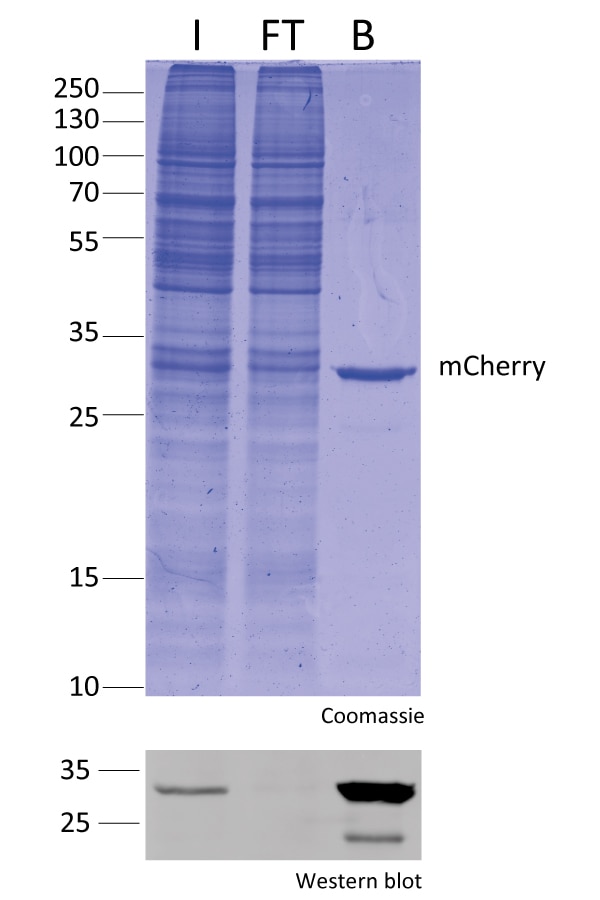
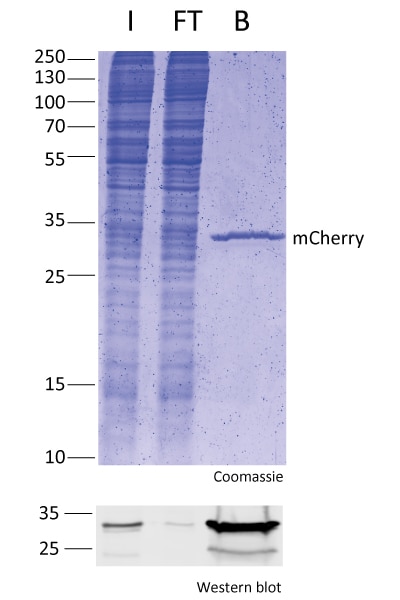
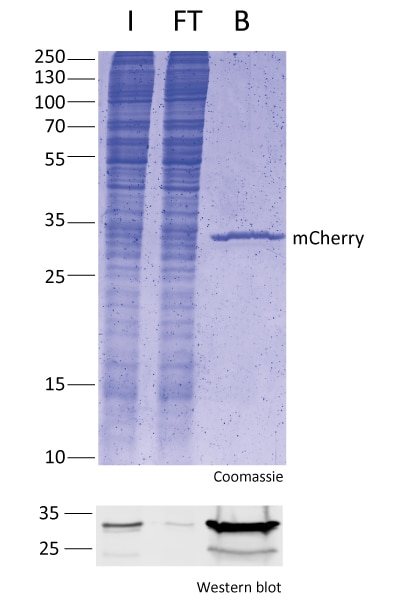
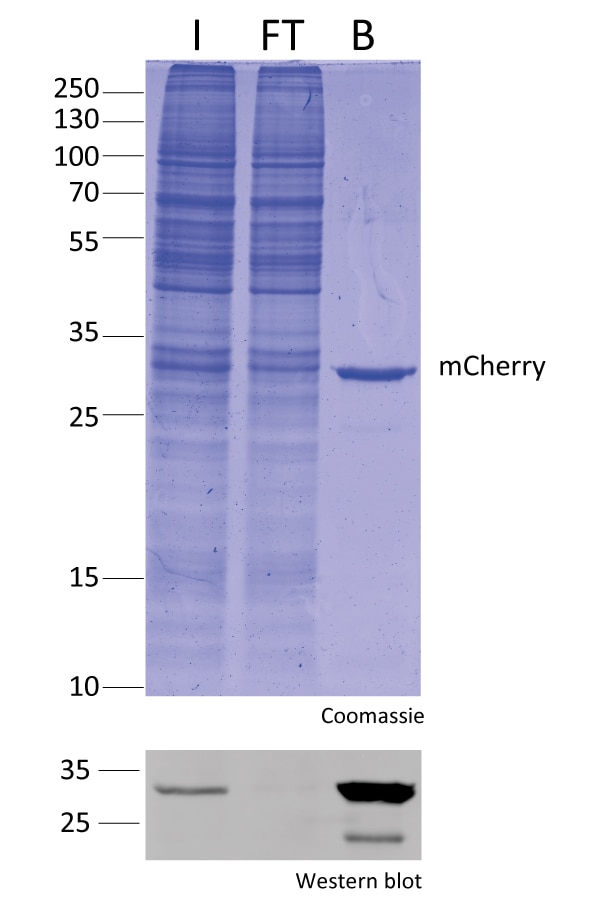
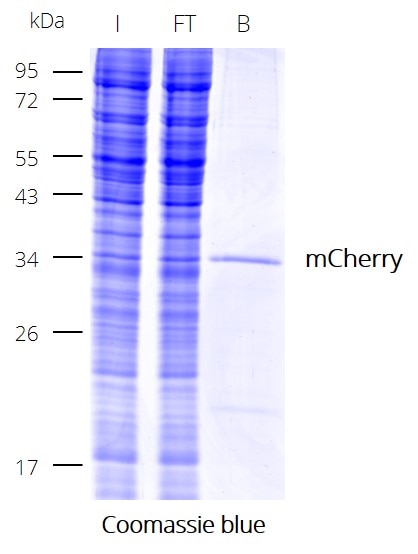
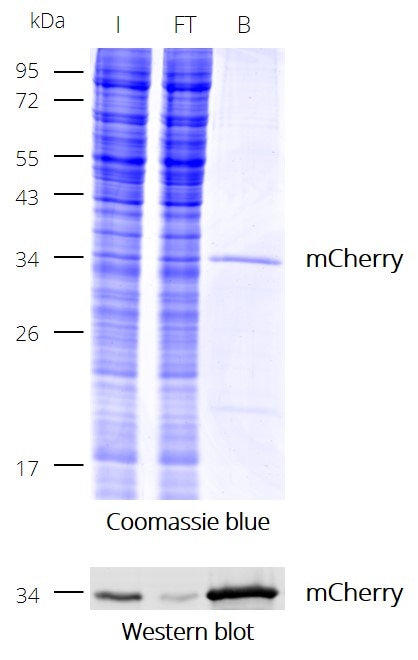
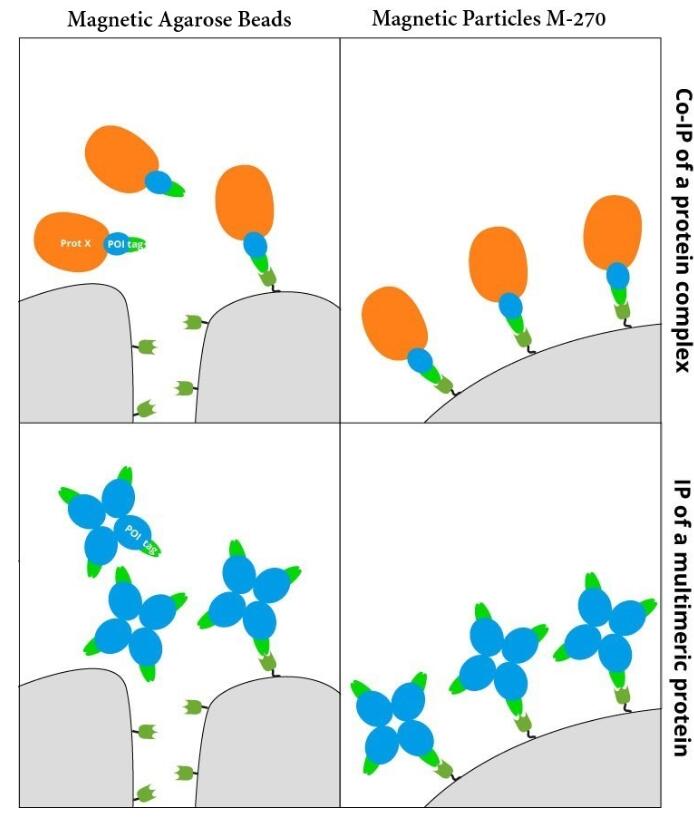
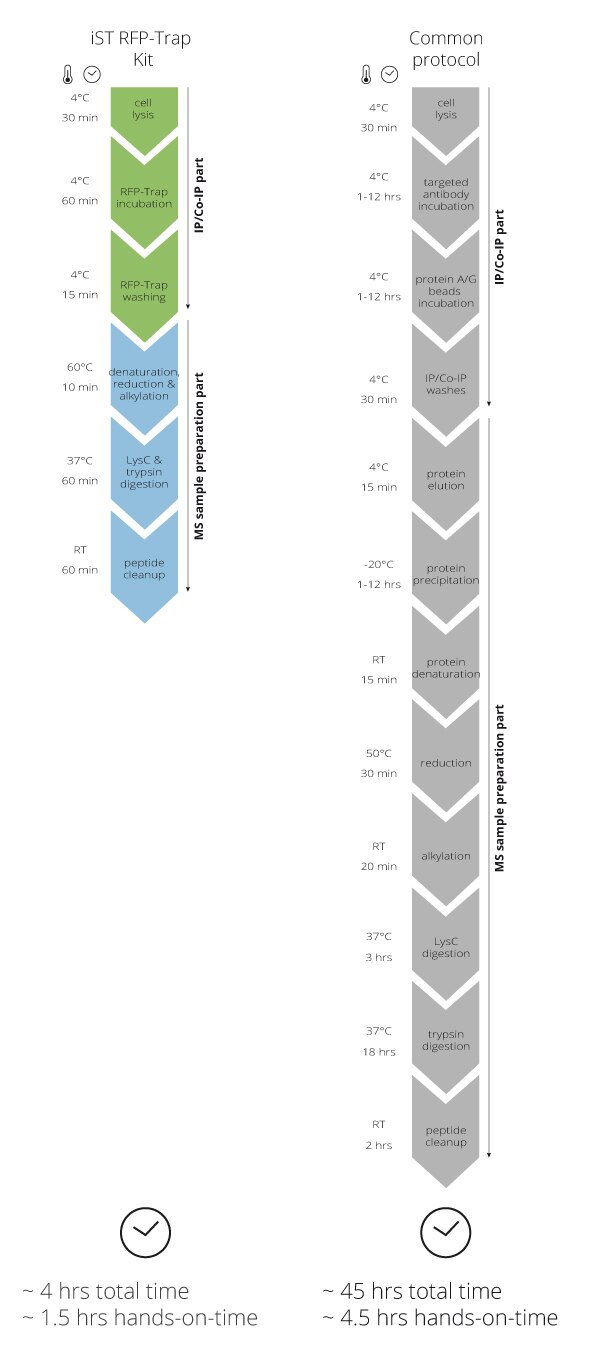
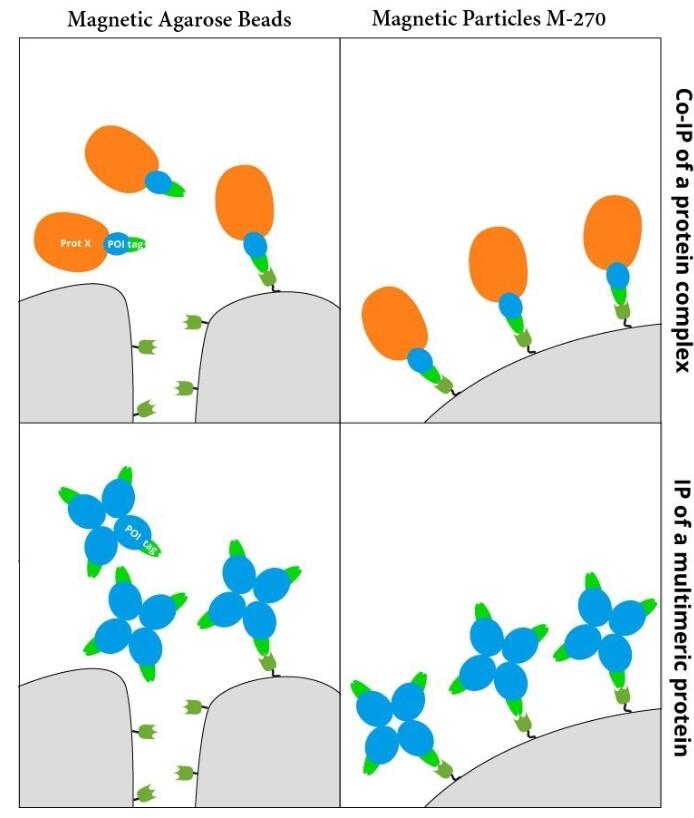
IP with RFP-Trap® NanobodiesThe ChromoTek RFP-Trap® is a ready-to-use affinity resin for immunoprecipitation (IP) of red fluorescent-fusion proteins like mCherry, dsRed, mRFP, and more. RFP-Trap consists of an anti-RFP Nanobody/ VHH coupled to agarose beads, magnetic agarose beads or solid magnetic particles. Specificity Applications |